Here’s something I’ve learned working with manufacturing facilities across three continents: most wipe-related problems don’t start with the wipe itself. They start with how that wipe has been treated before it ever touches a surface.
I’ve watched plants spend thousands on premium industrial wiping cloths only to see that investment wasted because someone left a case open near a loading dock, or stored wipes next to solvent containers, or let humidity fluctuate wildly in their supply room. The difference between a wipe that works and one that causes defects usually comes down to these details.
Let me share what we’ve found works. Not textbook theory—this comes from real facilities fixing real problems. Proper storage and handling can extend wipe life 2-3 times while keeping contamination control intact. When you’re managing a cleanroom or a manufacturing line, that’s money directly affecting your bottom line.
Why Wipe Storage Actually Matters in Manufacturing
Think about what a wipe is supposed to do: remove contaminants without adding new ones. That’s it. When wipes sit in the wrong environment, they start absorbing stuff from the air—dust, moisture, chemical vapors from nearby storage. They degrade. The packaging gets compromised. Then you bring these compromised wipes onto your production line.
I remember walking through a mid-sized electronics manufacturer where defect rates had been climbing for six months. Production manager was frustrated—same processes, same materials, same wipes. When we got to their storage area, the problem became pretty obvious. Cases of wipes stacked directly under a ventilation intake that was pulling in warehouse dust, and placed right next to their solvent dispensing station. The wipes were soaking up both airborne particulates and solvent fumes. Every time they wiped a surface, they were potentially leaving residue behind.

They reorganized their storage area—simple stuff, really—and their defect rate dropped 18% in three weeks. Same wipes, same people, same processes. Just better handling before the wipes ever reached the production floor.
What Actually Goes Wrong in Storage
From facility audits I’ve done, here are the contamination sources we find most often:
- Ambient particulates: Dust settles on packaging, gets in through compromised seals or when packages are left open.
- Moisture: High humidity makes wipes absorb water. Organic materials can grow microbes. Dry wipes don’t perform well when they’re damp.
- Chemicals: Storing wipes near volatile chemicals means solvents or other substances permeate the packaging.
- Temperature: Too hot accelerates material degradation. Too cold makes some materials brittle. Both affect performance.
- UV light: Direct sunlight breaks down synthetic fibers over time.
- Physical damage: Crushing cases with heavy stacks changes the material structure.
Real Impact on Your Operation
What happens when you use compromised wipes? It depends on what you’re making, but the consequences are always expensive:
- Cleanrooms: You introduce contaminants that your filtration system has spent millions trying to keep out. Particle counts spike, yields drop.
- Pharma: Cross-contamination risks can trigger recalls. Residues interfere with analytical testing. Regulators notice.
- Semiconductors: Microscopic contaminants kill wafers. One wafer lost to contamination costs more than proper storage for a year.
- Precision manufacturing: Residues affect tolerances, surface finishes, coating adhesion.
- Automotive painting: Contaminants in wipes cause paint defects that require rework or get rejected by customers.
The point here isn’t to make anyone paranoid. It’s to recognize that these are preventable problems. And preventing them starts with proper storage and handling.
Storage Conditions by Wipe Type
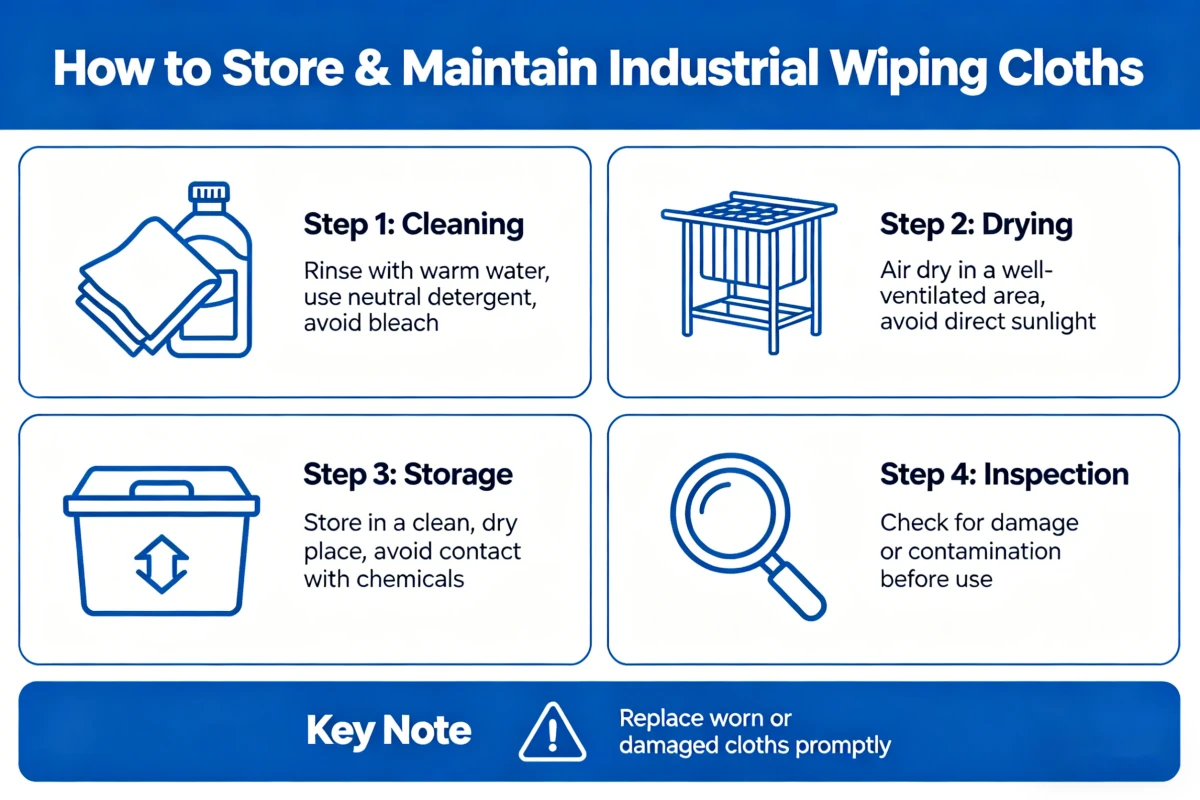
Not all wipes are the same, and they don’t all need the same storage conditions. Material matters. Application matters. Here’s what we’ve found works for different product types.
Dry Wipes: Polyester, Rayon, Cellulose Blends
Most dry industrial and cleanroom wipes share similar storage needs:
- Temperature: Keep it between 15-25°C (60-75°F). Extreme heat or cold changes material properties.
- Humidity: Aim for 30-60% relative humidity. Higher means moisture absorption and potential microbial growth. Too low creates static problems.
- Light: Keep wipes out of direct sunlight and away from UV sources. UV degrades synthetics over time.
- Location: Keep wipes away from chemical storage areas, exhaust vents, and anywhere particulates are being generated. Dedicate a specific area for cleanroom supplies.
- Position: Store cases flat. Don’t stack heavy objects on top—crushed material doesn’t perform the same.
- Rotation: Use oldest stock first. It sounds basic, but I’ve seen facilities lose stock because they used new before old.
Pre-Wetted Wipes
Pre-wetted wipes have extra considerations because they’re carrying solvent:
- Seal integrity: Check packaging seals carefully. A compromised seal lets solvent evaporate and contaminants in.
- Temperature: Often more sensitive than dry wipes. Follow manufacturer recommendations—some products specify tighter temperature ranges.
- Container position: Store upright as recommended. Some containers are designed specifically for upright storage to keep wipes properly saturated.
- Shelf life:Pre-wetted wipes have shorter shelf lives than dry wipes. Track expiration dates closely.
- Compatibility: Don’t store different pre-wetted wipe types together unless they contain identical solutions. Chemical mixing through cross-contamination is real.
Cleanroom-Specific Products
Wipes meant for cleanrooms need extra attention:
- Environment matching: Store in environments that match or exceed the ISO class where they’ll be used. No point using ISO Class 5 wipes that have been sitting in a Class 8 environment.
- Transfer procedures: When moving wipes into a cleanroom, follow proper gowning and pass-through procedures. Minimize time in transitional areas.
- Inner packaging: Many cleanroom wipes come double-bagged or sealed. Open only in the cleanroom environment where they’ll be used.
- Tacky mats: Place tacky mats outside storage areas and cleanroom entries to reduce tracked-in contamination.
Handling Best Practices That Actually Work
Storage doesn’t stop when you open the package. How wipes are handled during use affects their performance and your process cleanliness. We’ve developed these protocols from working with facilities across multiple industries.
Opening the Package Right
The moment you open a wipe package, contamination risk goes up. Here’s how we recommend handling it:
- Clean hands: Wash hands before opening wipe packages. Use gloves for cleanroom applications—non-negotiable.
- Clean environment: Open packages in appropriate environments. Don’t bring cleanroom wipes to the shop floor before opening.
- Inspect first: Look at wipes before using them. Discoloration, visible contamination, material degradation—these are red flags.
- Seal after use: Reseal packaging immediately after removing wipes. Don’t leave packages open between uses.
- Use oldest stock: Always start with the oldest package. Maintain FIFO rotation—first in, first out.
Taking Wipes Out Without Introducing Contamination
How you remove wipes matters, especially in cleanrooms:
- Minimize contact: Touch only the portion you won’t be using to clean. Don’t touch the entire wiping surface.
- Unfold carefully: Don’t shake or snap wipes—this generates particles. Unfold slowly and deliberately.
- Single use: Use each wipe for one area or surface. Don’t use the same wipe to clean multiple critical areas—cross-contamination is real.
- Proper disposal: Dispose according to facility protocols. Don’t leave used wipes near clean areas.
Cleanroom-Specific Handling
Cleanroom environments add another layer of requirements:
- Gowning compliance: Follow all gowning procedures before handling cleanroom wipes. Gloves are mandatory.
- Double gloving: Consider double gloving for critical applications. Change gloves between tasks.
- No skin contact: Never allow skin to contact the wiping surface. Skin oils and contaminants don’t belong on cleanroom surfaces.
- Controlled unfolding: Many cleanroom wipes are folded for a reason—unfold carefully to minimize particle generation.
Handling Mistakes We See Over and Over
In our facility audits, these mistakes show up constantly:
- Packaging left open: Operators leave wipe packages open between shifts, allowing environmental contamination.
- Improper mid-shift storage: Partially used packages stored in dirty areas or next to chemical containers.
- Wipe overuse: Wipes used beyond their capacity, spreading contaminants rather than removing them.
- Cross-contamination: Same wipe used for different areas without considering what transfers.
- Glove contamination: Gloves used to handle dirty equipment then used to retrieve clean wipes without changing.
- Improper disposal: Used wipes left near clean areas or in containers that let particles escape.
These aren’t just procedural issues—they’re contamination risks that affect product quality. Proper handling matters as much as proper storage.
Shelf Life and When to Replace Stock
All wiping cloths have finite shelf lives. The specifics vary by product type and storage conditions. Understanding these helps ensure you’re always using wipes that perform as intended.
Typical Shelf Lives by Product Type
| Product Type | Typical Shelf Life | Key Factors |
|---|---|---|
| Dry polyester wipes | 18-24 months | Storage conditions, packaging integrity |
| Dry cellulose wipes | 12-18 months | Humidity control, temperature |
| Pre-wetted solvent wipes | 6-12 months | Seal integrity, temperature, evaporation |
| Pre-wetted alcohol wipes | 6-12 months | Alcohol evaporation, seal condition |
| Anti-static wipes | 12-18 months | Treatment degradation, storage conditions |
| Specialized cleanroom wipes | 12-24 months | ISO class requirements, environment |
Signs Your Wipes Have Degraded
Before using wipes—especially if they’ve been in storage a while—check for these warning signs:
- Discoloration: Yellowing, darkening, or uneven coloration can indicate UV degradation or chemical exposure.
- Odor changes: Unusual odors suggest chemical contamination or microbial growth.
- Texture changes: Wipes becoming brittle, stiff, or unusually soft signal material breakdown.
- Visible contamination: Any visible particles, stains, or foreign materials.
- Moisture issues: Dry wipes feeling damp, or pre-wetted wipes showing signs of drying or solvent separation.
- Seal compromise: Packaging that’s torn, punctured, or shows seal failure.
When to Replace Your Stock
Establish clear replacement criteria based on your applications:
- Expiration dates: Respect manufacturer expiration dates, especially for pre-wetted products.
- Storage incidents: Any wipe stock exposed to extreme conditions or potential contamination should be evaluated for replacement.
- Performance testing: For critical applications, consider periodic testing of stored wipes to verify they still meet specifications.
- Package condition: Damaged or compromised packages shouldn’t be used in critical applications.
Inventory Management That Works
Good inventory management ensures optimal use of your wipe stock:
- FIFO rotation: Always use oldest stock first. Track lots and monitor expiration dates.
- Regular audits: Schedule inventory audits to check package condition and expiration dates.
- Clear labeling: Label stock with receipt dates and lot numbers for easy tracking.
- Separate locations: Store different wipe types in designated areas to prevent mix-ups and cross-contamination.
- Training documentation: Document handling procedures and train everyone who handles wipes.
Why Our Team Understands Your Contamination Challenges
Proper wipe storage and handling isn’t just about following a checklist—it’s about understanding contamination control principles. At WIPESTAR, our team has decades of combined experience across multiple industries.
Our Contamination Control Specialists
Our technical team includes specialists with backgrounds in semiconductor manufacturing, pharmaceutical production, aerospace, and precision engineering. We understand cleanroom operations from the inside because we’ve worked there. This hands-on experience informs everything we do—from product selection to customer support.
When you work with WIPESTAR, you’re not just buying wipes. You’re accessing contamination control expertise that helps optimize your entire cleaning process. We help customers audit procedures, identify contamination sources, and implement best practices that improve yield and reduce defects.
Industry-Specific Experience
Different industries face different contamination challenges. Our team has worked in:
- Semiconductor fabrication: From Class 1 to Class 100,000 cleanrooms, we understand particle control at every level.
- Pharmaceutical manufacturing: We’re familiar with GMP requirements, cross-contamination prevention, and regulatory expectations.
- Medical device production: We understand balancing cleanliness requirements with production efficiency.
- Aerospace manufacturing: We’ve helped facilities with precision cleaning for critical aerospace components.
- Automotive painting: We’ve worked with paint shops reducing defect rates through improved wiping procedures.
Training and Support
Knowledge transfer is part of our service. We offer training programs for facilities that want to improve their contamination control practices. From basic wipe handling to advanced cleanroom protocols, we can help your team develop the expertise they need.
Learn more about our team and how we can support your contamination control efforts.
Storage and Handling Checklist
Use this checklist to ensure proper wipe storage and handling in your facility:
Storage Environment
- Temperature maintained at 15-25°C (60-75°F)
- Humidity controlled at 30-60% relative humidity
- No direct sunlight or UV exposure
- Located away from chemical storage areas
- Dedicated storage area for cleanroom supplies
- Free from dust sources and ventilation intakes
- Cases stored flat, not stacked with heavy objects
Package Management
- First-in, first-out inventory rotation
- Clear lot tracking and expiration date monitoring
- Regular package condition inspections
- Damaged packages removed from inventory
- Different wipe types stored separately
- Pre-wetted wipe seals inspected regularly
Opening and Use
- Hands washed before opening packages
- Appropriate gloves used (required for cleanroom applications)
- Packages opened only in appropriate environments
- Wipes inspected before use
- Packaging resealed immediately after use
- Wipes removed with minimal contact
- Wipes unfolded carefully to minimize particles
Cleanroom-Specific
- Storage environment matches cleanroom ISO class
- Proper gowning procedures followed
- Pass-through protocols used when entering cleanroom
- Inner packaging opened only in cleanroom
- Double gloving used for critical applications
- No skin contact with wipe surfaces
Documentation and Training
- Written procedures for wipe storage and handling
- All personnel trained on procedures
- Training records maintained
- Regular procedure reviews and updates
- Incident reporting for storage or handling issues
Frequently Asked Questions
Getting Started with Improved Wipe Management
If you’re experiencing contamination issues that might trace back to wipe storage or handling, or if you simply want to optimize your current procedures, we’re here to help. Our contamination control specialists can review your practices, identify opportunities for improvement, and provide recommendations tailored to your specific application.
Proper wipe storage and handling isn’t complicated—it just requires attention to detail and consistent procedures. The payoff comes in improved product quality, reduced defects, and better use of your consumable investment. Start with this checklist, reach out if you need expert guidance, and you’ll see the difference that proper wipe management makes.
























