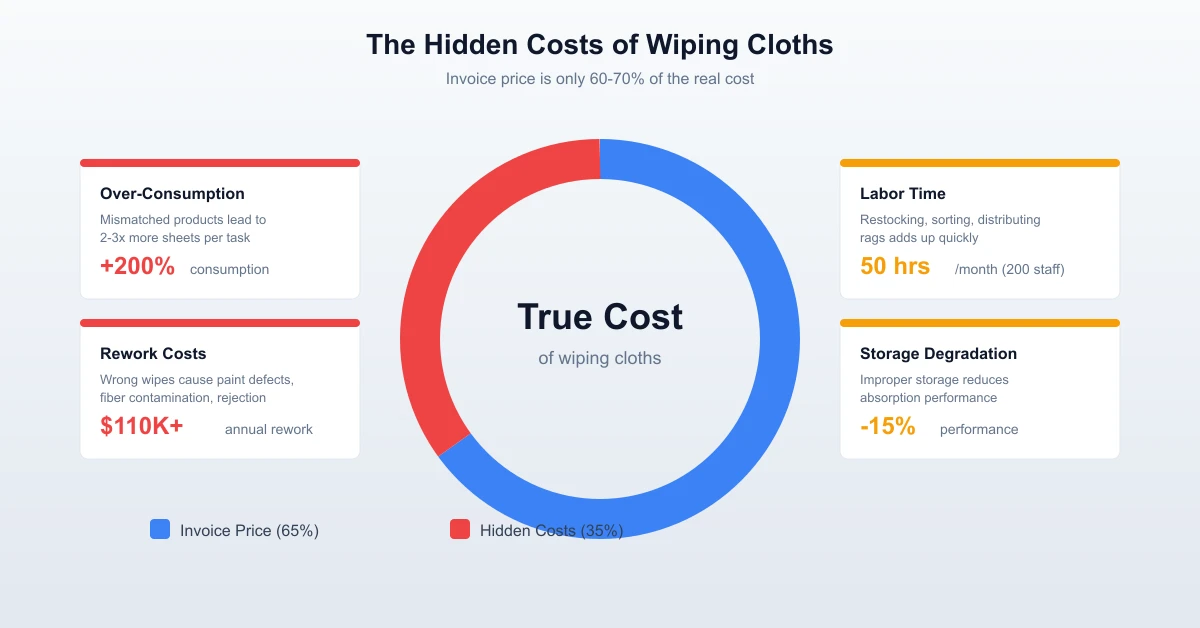
Over the past decade, I’ve walked through dozens of pharmaceutical production floors — from aseptic filling suites in Shenzhen to API synthesis workshops in Jiangsu. One thing consistently surprises me: how often wipe selection is treated as an afterthought. Facilities invest millions in HVAC, HEPA filtration, and gowning protocols, then grab whatever lint-free wipe happens to be on the shelf. That’s a gap we see close up at WIPESTAR, and it’s exactly why I wrote this guide.
If you’re managing a pharmaceutical cleanroom — whether it’s ISO Class 5 aseptic filling or ISO Class 7 packaging — the wipe you choose directly impacts your contamination control outcomes. Wrong material, wrong particle count, wrong extractables profile, and you’re introducing the very contaminants you’re trying to eliminate.
This isn’t a product pitch. It’s the conversation I have with pharma procurement teams every week, distilled into something you can act on.

Why Pharmaceutical Cleanroom Wiping Is Different
I hear this a lot: “A wipe is a wipe — why does pharma need something special?” Fair question. Here’s the honest answer: pharmaceutical manufacturing environments have constraints that most industrial settings simply don’t.
First, there’s the extractables and leachables (E&L) concern. Any chemical that migrates from the wipe into your product — even at parts-per-billion levels — can compromise drug safety. In a CNC shop, a trace of isopropanol residue from a wipe doesn’t matter. In an injectable drug filling line, it can trigger a batch rejection.
Second, bioburden control. Pharma cleanrooms need wipes that don’t introduce microbial contaminants. Standard industrial wipes aren’t manufactured or packaged to meet those requirements.
Third, regulatory traceability. FDA 21 CFR Part 211, EU GMP Annex 1, and ISO 14644 all touch on cleaning materials in controlled environments. Your wipes need documentation — certificates of analysis, ROHS compliance, particle count data — that holds up under audit.
This is fundamentally different from wiping down a milling machine. If you’re sourcing cleanroom consumables for pharma, the selection criteria shift dramatically.
Understanding ISO Classes: Which Wipe for Which Environment
Not every pharma cleanroom needs the same wipe. I’ve seen facilities over-specify (wasting budget on ISO Class 5 wipes in an ISO Class 8 corridor) and under-specify (using general-purpose wipes in an aseptic core). Both are problems.
Here’s how I break it down for clients:
ISO Class 5 (Aseptic Core / Filling Rooms)
This is the most critical zone. You need wipes with extremely low particle generation, validated extractables profiles, and often pre-wetted formats to minimize manual handling. Sterile, double-bagged packaging is typically required. Materials like continuous-filament polyester or knitted nylon are standard here.
ISO Class 6–7 (Formulation, Compounding, Weighing)
Slightly relaxed, but still demanding. Lint-free construction is non-negotiable. I recommend ultrasoft surface preparation wipes for sensitive equipment, and heavier-duty options like the W3501 surface preparation wipes for general bench and equipment cleaning where abrasion resistance matters.
ISO Class 8 (Packaging, Gowning, corridors)
General-purpose cleanroom wipes work well here. Cost efficiency matters more since usage volumes are higher. A good universal wiping cloth handles most tasks — surface wiping, spill cleanup, equipment maintenance — without the premium pricing of ISO Class 5 consumables.
The mistake I see most often? Using the same wipe across all zones. It either blows the budget or introduces contamination. Match the wipe to the ISO class — that’s the first rule.

Cleanroom Wipe Materials: What Works in Pharma
Material selection is where most procurement decisions go wrong. Let me walk through the options we work with most frequently in pharmaceutical applications:
Continuous-Filament Polyester
The gold standard for ISO Class 5 environments. The fibers are extruded as a single continuous strand — no short fibers to shed. Particle generation is extremely low. Ideal for aseptic filling, sterile compounding, and any surface that contacts the drug product directly. The tradeoff is cost; these are premium wipes.
Knitted Nylon
Excellent abrasion resistance makes knitted nylon a strong choice for cleaning equipment with rough surfaces — stainless steel tanks, mixer blades, tablet press tooling. Absorbency is moderate, so it pairs well with pre-wetted formats.
Wood Pulp / Polyester Blends
The workhorse for ISO Class 7–8 environments. These blends balance absorbency, strength, and cost. Our wiping cloths lineup includes several wood pulp/polypropylene options that pharma clients use daily for general surface cleaning, spill response, and equipment wipe-down in less critical zones.
Microfiber
Growing in pharma applications, especially for glass and polished stainless surfaces. The split-fiber structure traps particles rather than pushing them around. However, microfiber can be problematic with certain solvents — always validate chemical compatibility before committing.
When I sit down with a pharma client, the first question isn’t “which wipe is best?” — it’s “what are you cleaning, in which ISO zone, with what chemicals?” The answer determines everything. Our cleanroom wiper selection guide goes deeper on material science if you want the technical breakdown.
5 Contamination Risks That Poor Wipe Selection Creates
I’ve audited enough pharma facilities to see the same problems repeat. Here are the five contamination risks that trace back to wipe selection — and they’re more common than you’d think:
1. Particle Shedding from Low-Grade Wipes
Cheaper wipes use short-cut fibers that shed under pressure. In a cleanroom, those fibers become airborne particulates. For oral solid dosage, it’s a nuisance. For injectables, it’s a recall waiting to happen.
2. Extractable Chemicals Leaching into Product
Wipes manufactured with binders, surfactants, or antimicrobial agents can release trace chemicals when exposed to solvents. If your wipe isn’t validated for E&L, you’re running blind.
3. Microbial Introduction from Non-Sterile Packaging
Standard industrial wipes aren’t packaged to prevent bioburden. Opening a non-sterile bag in an aseptic core defeats the purpose of your entire gowning and disinfection protocol.
4. Cross-Contamination from Reuse
Some wipes are marketed as reusable but harbor residual contamination after rinsing. In pharma, single-use is almost always the safer call for critical zones.
5. Solvent Incompatibility Causing Residue
Using a wipe that breaks down in IPA or quaternary ammonium compounds leaves residue on surfaces — exactly what you’re trying to remove. Always test wipe-solvent compatibility before facility-wide rollout.
Our Selection Checklist: How We Evaluate Wipes for Pharma Clients
When a pharmaceutical manufacturer contacts us — whether it’s a CDMO in Suzhou or a generics producer in Hyderabad — we walk through the same evaluation framework. Here’s the checklist, simplified:
- Define the ISO zone — Where will the wipe be used? This determines acceptable particle counts and cleanliness levels.
- Identify the target contaminant — Oil, solvent, bioburden, particulate, or adhesive residue? Each demands different wipe properties.
- List chemical exposures — What disinfectants, solvents, or cleaning agents will contact the wipe? Material compatibility is critical.
- Confirm sterility requirements — Aseptic zones require sterile, double-bagged wipes. Non-aseptic zones can use standard packaging.
- Review regulatory documentation — Do you need COA, ROHS certification, or USP <797> compliance documentation? We provide all of these on request.
- Request samples and validate — We never push bulk orders without on-site trials. Send us your specs, and we’ll match the right wipe from our product catalog.
This process takes a bit more time upfront, but it eliminates the costly cycle of trial-and-error that plagues so many pharma facilities.
Real Scenarios from Our Pharma Clients
I want to share a couple of anonymized situations that illustrate why this matters in practice:
Case 1: Injectable Filling Line — Particle Excursion
A contract manufacturer was seeing intermittent particle excursions in their ISO Class 5 filling room. After investigating their gowning materials, disinfectants, and HEPA integrity (all clean), we traced the issue to the wipes they were using on the filling machine. The wipes were general-purpose polyester with unacceptable particle shedding for aseptic applications. We switched them to a continuous-filament polyester wipe — same dimensions, same dispensing format — and the excursions stopped within two production cycles.
Case 2: Tablet Coating Room — Solvent Residue
A generics producer was cleaning their tablet coating pans with IPA-soaked wipes that left a visible residue. The wipes were cellulose-based, and the IPA was dissolving binders in the wipe material. We recommended a synthetic blend wipe with validated solvent compatibility. The residue issue resolved immediately, and they avoided a costly equipment modification.
These aren’t edge cases. They’re Tuesday morning calls for our team.
Why Our Team Understands Pharma Cleaning
I think it’s worth explaining why WIPESTAR is positioned to advise on this topic — not because I want to sell you something, but because credibility matters when you’re trusting someone with your contamination control strategy.
Our Sales Director, Ethan, has spent over 20 years in the industrial wiping cloth and cleanroom consumables industry. He’s led our global sales team through hundreds of pharma and semiconductor facility audits, and he built our product evaluation methodology from the ground up.
Carolina, our Product Specialist, works directly with production technicians and quality teams at client facilities. She doesn’t just spec products — she visits production floors, talks to the people using the wipes, and feeds that real-world data back into our product development. Her understanding of how cleanroom consumables perform under actual production stress (not just lab conditions) is something I genuinely value.
Juan, our Purification Industry Specialist, brings deep expertise in cleanroom design and purification process equipment. When a pharma client needs guidance on integrating wipe selection into their broader contamination control strategy — HVAC, gowning protocols, disinfection cycles — Juan is the one who connects those dots.
Yunyun, our ESD Project Manager, may seem like an odd fit for a pharma article, but her experience in static control technology is directly relevant. Electrostatic discharge can attract and hold airborne particles on surfaces — a contamination vector that many pharma teams overlook when evaluating wipe materials.
And then there’s Guan, who spent 4.5 years in the semiconductor industry before joining us. Semiconductor cleanrooms operate at ISO Class 1–3 — environments even more stringent than pharma. His perspective on wipe performance at extreme cleanliness levels has been invaluable for our pharmaceutical clients pushing into ISO Class 5 territory.
This isn’t a team that learned cleanroom consumables from a catalog. We’ve been in the rooms, under the hoods, and on the production floors. That experience is what you’re tapping into when you work with our team.
Frequently Asked Questions
Can I use the same cleanroom wipes across all ISO zones in my pharma facility?
Technically you can, but it’s not cost-effective and often not compliant. ISO Class 5 zones require sterile, ultra-low-particulate wipes. ISO Class 8 zones can use standard cleanroom wipes. Using ISO Class 5 wipes everywhere inflates costs unnecessarily; using ISO Class 8 wipes in aseptic cores risks contamination. Match the wipe to the zone.
What certifications should I look for when sourcing pharmaceutical cleanroom wipes?
At minimum: ROHS compliance, Certificate of Analysis (COA) with particle count data, and material safety data. For aseptic applications, add sterility validation (gamma irradiation or autoclave-compatible packaging). If your facility is audited under EU GMP Annex 1 or FDA 21 CFR Part 211, documentation traceability is non-negotiable. We provide all standard certifications and can supply additional validation documentation on request — just reach out to our team.
How do I know if a wipe is compatible with my facility’s disinfectants?
Request a chemical compatibility chart from your wipe supplier. Common pharma disinfectants — quaternary ammonium compounds, hydrogen peroxide, peracetic acid, IPA — interact differently with wipe materials. We test all our cleanroom wipes against standard pharma disinfectant formulations and can share compatibility data specific to your cleaning protocol.
What’s the difference between pre-wetted and dry cleanroom wipes for pharma?
Pre-wetted wipes come saturated with a validated cleaning solution (usually IPA/water or DI water). They reduce the risk of over-pouring solvents, ensure consistent wetness, and minimize touch contamination. Dry wipes offer flexibility — you control the solvent type and volume. For aseptic cores, pre-wetted sterile wipes are increasingly the standard. For general pharma cleaning, dry wipes paired with controlled solvent dispensing work well.
Does WIPESTAR offer custom packaging for pharmaceutical cleanroom wipes?
Yes. We provide custom sizing, private-label packaging, sterile double-bagging, and lot-traceable labeling. If your facility has specific packaging requirements — say, compatible with your dispensing systems or matching your gowning protocol — talk to our sales team about OEM options. We’ve built custom configurations for CDMOs, generics manufacturers, and biotech startups alike.
Need Guidance on Cleanroom Wipe Selection for Your Pharma Facility?
WIPESTAR supplies pharmaceutical-grade cleanroom wipes direct from our Shenzhen facility, with full documentation, bulk pricing, and global shipping. Every material is ISO-certified and ROHS compliant.
Our team — Ethan, Vicky, Lee, Carolina, Guan, Juan, and Yunyun — has collectively supported contamination control programs at pharmaceutical, biotech, and semiconductor facilities across Asia, Europe, and North America. If you’re evaluating wipe options for a new facility, a line expansion, or simply trying to fix a recurring contamination issue, we’d rather have the conversation upfront than hear about the problem after a batch fails.
Vicky:Foreign Trade Sales Supervisor, WIPESTAR Industrial Wipes
Vicky manages overseas client relationships for pharmaceutical and manufacturing accounts at WIPESTAR. She coordinates between our product specialists, quality team, and international clients to ensure cleanroom consumable solutions meet both regulatory requirements and operational realities.
This article is for industrial contamination control guidance only, not medical advice.-WIPESTAR